|
These guys are the mass number, and these guys just to read the fact that chlorine has 17 protons this is the atomic number. Another way to symbolize isotopes you might see it looking like this with a dash and a number behind it or you may see something looking like this. These guys are isotopes of each other meaning they have different number of neutrons. So in this case we know chlorine has 17 protons that's always a given and we have x number of neutrons in this case it will be 18 neutrons.įor chlorine 37 we know that the 17 protons plus x number of neutrons equals 37 in this case we have 20 neutrons. So in this case 35 is the number of protons plus the number of neutrons equals 35. So how do we denote when something isn't an isotope? Well there are two ways to denote it, we can either have the symbol with the mass number behind it, this is the mass number, this is the number of protons plus neutrons. But pretty much the chemical reactivity or the chemical properties are identical. 
And I change when talking about radio activity or something along those lines which we'll get into which is another whole topic amongst itself. They don't change that much at all when you gain or lose neutrons. Now the chemical reactivity and the chemical properties of isotopes are pretty much identical. That's when an atom has the same number of protons but different number of neutrons. If we change number of neutrons we're going to call those guys isotopes. And the ions can be positively or negatively charged depending on if we've gained or lose electrons within an element. We'd call these guys ions they're charged particles. If we're going to say okay it's going to gain an electron, we're now going to change make it Cl minus it gained an electron, meaning that it has one more negatively charged particle than it does positively charged particle. So let's say chlorine if we wanted to know that it's neutral, we're just going to say Cl with nothing on it. If you change the number of electrons, all you're changing is the charge or the electrical charge of that particular element. This actually gives the atom its identity, this does not change. If you lose a proton and you have 16 protons you're actually going to change it and it's now going to be sulphur. If you add a proton making it 18 you're actually going to change the identity of that element making it argon. so in this case you're talking about chlorine it will charge like we said 17 protons. If the number of protons changes within the atom, you're actually going to change the identity of that particular atom. Okay so these things are actually going to change for subatomic particles protons, neutrons, electrons are going to change. That gives an element mass, neutrons and protons you're like "I don't really understand because this is a decimal but we know that neutrons are 1amu and protons are also 1amu so where is this in a decimal?" Well this is the average atomic mass and what that means is we'll actually talk about what that means and how we calculate that and what that actually entails in just a second. What gives an element or an atom mass? Well the number of neutrons in the element and the number of protons. The last thing that you might see on the periodic table that's important right now would be this number 35.453 what does that mean? That's the average atomic mass that's measured in atomic mass units. 
But if it has two letters in it make sure that the second letter is lower cased. So anytime that you have a chemical symbol that has two letters in it, not all of them has two letters. This is completely incorrect, to me as a chemist when I see CL like that one element C and the second element L and I know that you meant C and a little l. So one thing I see a lot that students do is instead of making it a lower case l the second one kids a lot of times just keep them both in capital letters. Cl is the chemical symbol or how we'd see it maybe in chemical formula or something like that, a way to make it shorter. 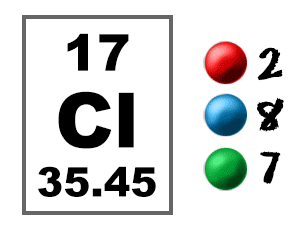
Is the number 17, so chlorine has 17 protons. So chlorine is it's chemical name and might not have it on your periodic table it really depends on what periodic table you're looking at but that's the official chemical name the number 17 will be on there also and that indicates the atomic number, it always is the number of protons in that particular element. So here is the element that you might see chlorine on the periodic table and I just kind of brew it up for you so we can talk about what these numbers mean. But I'm going to help you go through the periodic table and tell you what exactly it is that you're looking at. We're going to talk about the atomic number and isotopes on the periodic table you might see a lot of things that are confusing to you.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
